Discover how stress impacts your gut health, including its impact on microbiome diversity and digestion.
If you've ever felt "butterflies in your stomach" when you were anxious, you'll know that stress can play out in the gut. But what happens when you enter a fight-or-flight state, and how does a psychological response translate to digestive symptoms? Keep reading to find out.
Table of contents
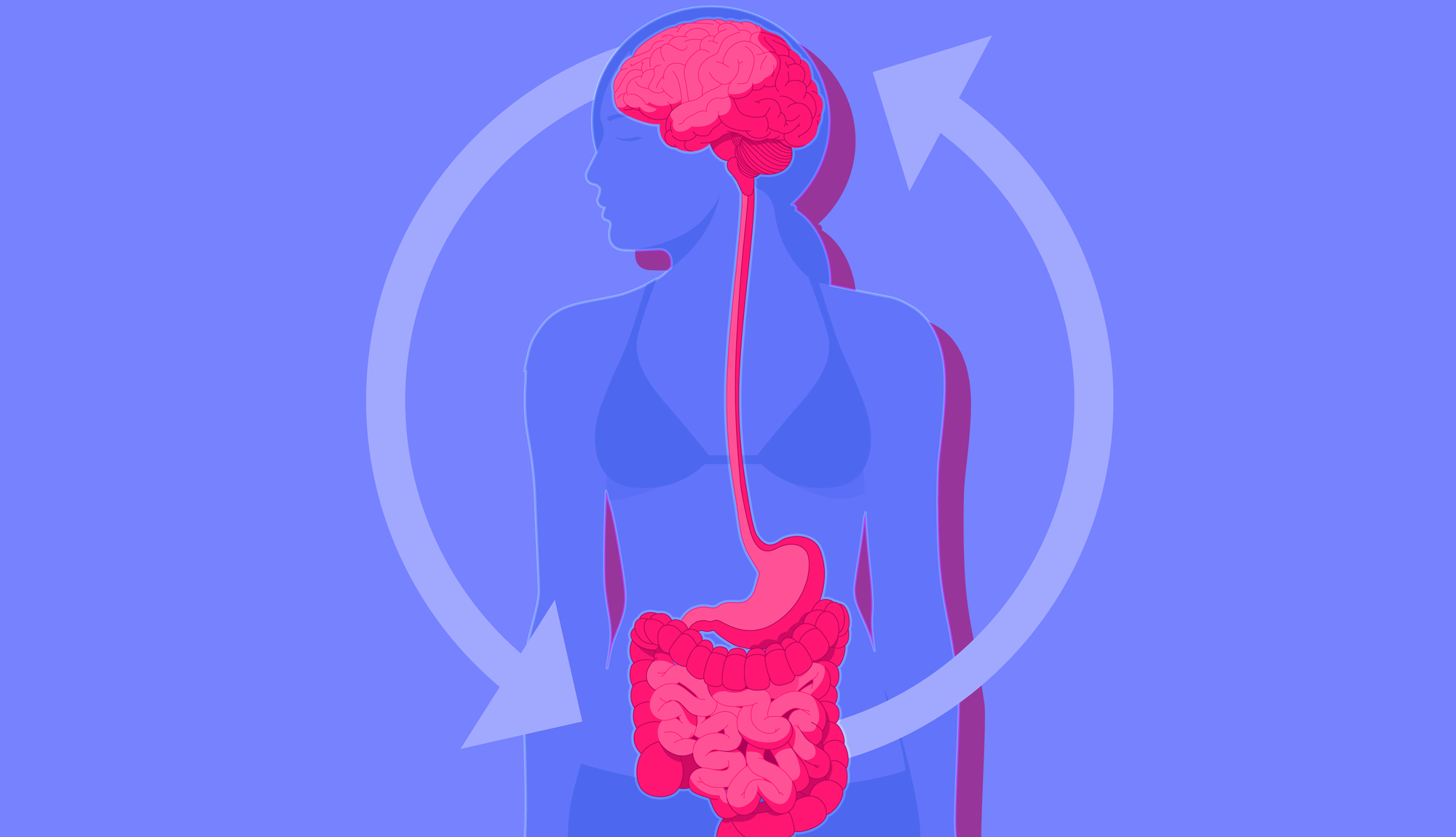
The gut-brain axis
In Ancient Egypt, those responsible for embalming bodies would carefully preserve all the organs in a mummies body, except the brain.
When it came to the 3 pounds of grey mass responsible for thought, memory, movement and speech, they scooped it out through the nose with a sharp instrument.
We laugh today at their disregard for this remarkably complex human organ, but for many years, we have all neglected a second brain that resides in the human gut, known as the enteric nervous system.
Sandwiched between our epithelial cells in the colon and small intestine, this complex network of nerves and neurons is in charge of all gastrointestinal processes, managing blood flow, muscle contractions and the release of digestive fluids.
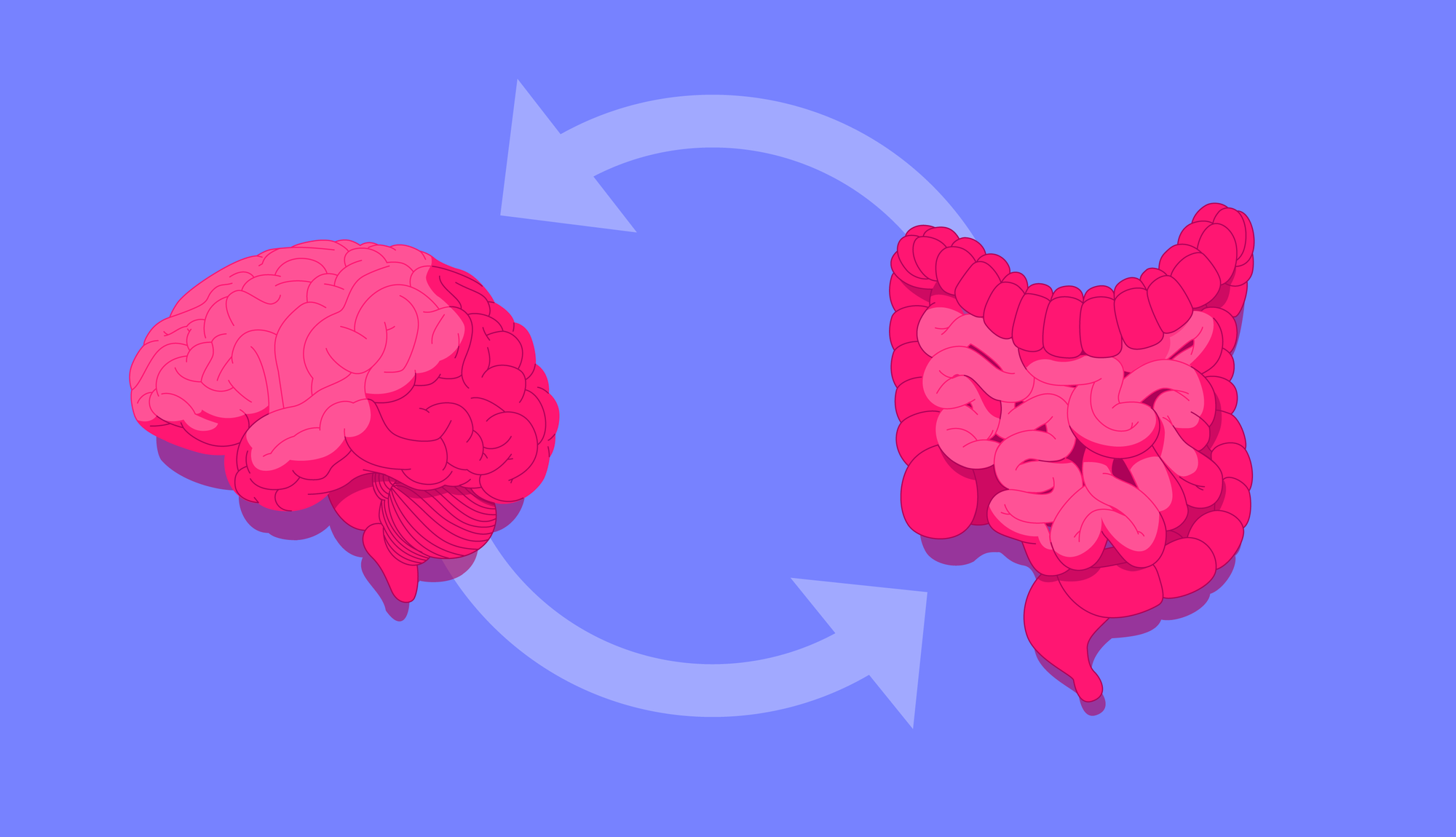
Researchers refer to this link as the gut-brain axis, a two-way communication network connecting the cognitive and emotional centres of the brain with our intestinal functions.
Humans seem to know this intuitively, as reflected in our language: we have a “gut feeling” or “butterflies in the stomach” when we are nervous, which is to say that our emotions are felt in the gut.
The gut-brain axis goes a long way to explaining why those with Irritable Bowel Syndrome exhibit higher rates of depression and anxiety.
In light of this connection, it would make sense that psychological interventions for anxiety or depression could alleviate IBS symptoms, and that’s what emerging research suggests.
In some studies, GDH has been shown as effective as dietary interventions such as a low-FODMAP plan. So how do these two ostensibly separate organs stay in contact?
In short, the enteric nervous system communicates with the brain via multiple pathways, including the vagus nerve, autonomic and central nervous system.
Considering that the gut and brain are so closely connected, it should come as no surprise that stress can lead to a range of gastrointestinal disorders and symptoms, as we shall explore.
☝DID YOU KNOW?☝ Even thinking about food can trigger the gut to secrete digestive enzymes in preparation for a meal.
How Stress Affects The Gut

In scientific research, our automatic stress response is known as fight-or-flight, and it serves an important evolutionary purpose. Once upon a time, the fight-or-flight response helped your Stone Age ancestors to avoid being a lion’s dinner.
While few of us have to worry about fleeing ravenous lions anymore, the physiological response remains and is triggered in response to any situation we deem threatening, be that a mid-term exam or a presentation.
When you encounter a situation you perceive as stressful, the sympathetic nervous system is activated and initiates an acute stress response.
As a result, the body ramps up its production of cortisol, which induces numerous changes throughout the body, including the gut.
Firstly, stress signals travel along the gut-brain axis, instructing the gut to direct energy elsewhere in the body; if you need to flee from a ravenous predator, digestion is a waste of energy.
As a result, blood is diverted away from the intestines towards your limbs, slowing digestion and potentially causing sudden evacuation (diarrhoea, to you and me).
Combine this with stress-related muscle spasms, and stress can be a recipe for gut issues. Worse still, stress increases visceral hypersensitivity, meaning you feel an increased pain response.
Adding to this cocktail of digestive woes, stress causes the gut to produce less mucus, a protective layer which coats the bowel wall.
It should come as little surprise that chronic stress is associated with increased intestinal permeability, colloquially known as “leaky gut”. In turn, this can cause low-grade chronic inflammation.
The stress response is perfectly natural and harmless in the short term (acute), but issues can arise if we remain in a prolonged state of stress (chronic).
Prolonged stress is linked with an increased risk for a range of disorders, including obesity, heart disease, diabetes and depression.
Additionally, chronic stress has long been known to contribute to the onset and worsening of symptoms in multiple GI disorders, including IBS and Inflammatory Bowel Diseases.
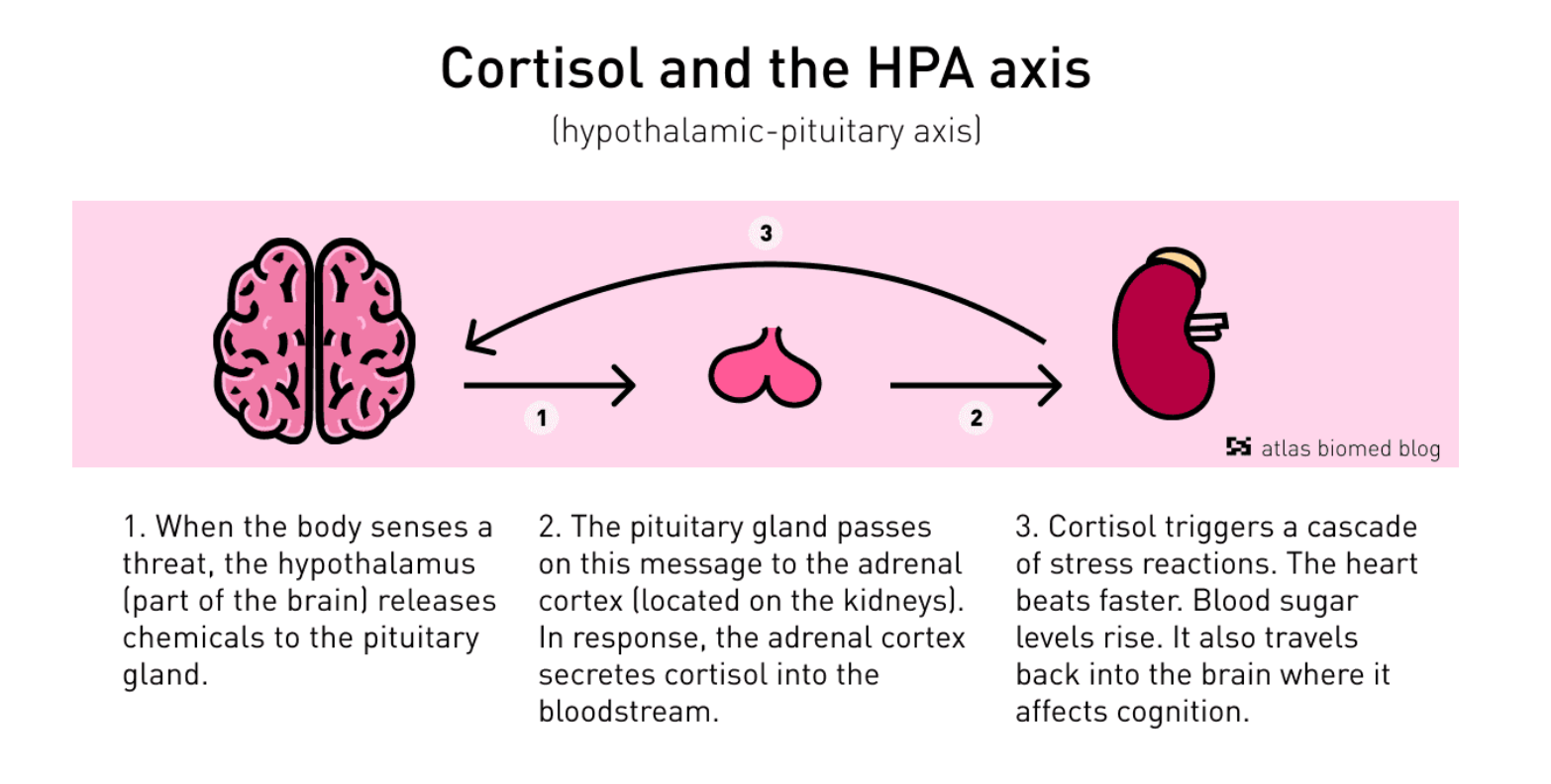
Prolonged stress and early-life trauma are thought to cause dysregulation in the hypothalamic-pituitary-adrenal axis (HPA), leading to increased production of Corticotropin-Releasing Hormones (CRH), a precursor for cortisol.
The HPA axis regulates the body’s stress response, thereby determining the extent of the fight-or-flight response.
When it goes wrong, it can leave someone stuck in an acute stress phase. Interestingly, HPA dysfunction is widely observed in numerous mental health conditions.
Stress and the microbiome

Observational studies have shown that psychological stress is associated with an altered microbiome in humans, though a cause and effect relationship has not been established.
Despite this, researchers postulate numerous plausible mechanisms by which stress may disrupt microbiome diversity, both directly and indirectly.
Firstly, exposure to stress can trigger comfort eating and cravings for ultra-processed foods high in sugar, fat, and salt. A diet enriched in these foods can disrupt microbiome diversity and lead to dysbiosis.
Beyond stress-related dietary changes, it has been suggested that the physiological changes caused by stress impact bacterial populations.
According to this hypothesis, altered gut motility and reduced mucus production essentially change the weather in your gut, allowing robust pathological bacteria to reproduce at the expense of beneficial or commensal microbes.
Another hypothesis is that noradrenaline and norepinephrine (NE) hormones may fuel changes in the gut microbiome. The idea is supported by animal studies in which NE has been observed to alter the gene expression of certain bacteria.
Research suggests that the link between our microbiome and stress is bi-directional, with several lines of evidence indicating that our gut microbiota mediates the stress response:
A groundbreaking study in 2004 further implicated the microbiome in stress responsiveness. In short, researchers managed to reverse the exaggerated HPA response by implanting germ-free mice with specific bifidobacterium bacteria.
Lastly, germ-free mice transplanted with faeces from depressed patients exhibit increased depressive symptoms and anxiety-like behaviour, suggesting our gut bugs can influence mood.
Key takeaways
- The gut-brain axis is a two-way communication network connecting the cognitive and emotional centres of the brain with our intestinal functions.
- The fight-or-flight response is an automatic physiological response to perceived or actual danger.
- When you are stressed, the body ramps up its production of cortisol, which induces numerous changes throughout the body, including the gut.
- The acute stress response slows digestion, potentially causing diarrhoea. What’s more, stress can trigger painful muscle contractions in the gut and increase pain sensitivity.
- Stress can drive and augment numerous gastrointestinal issues, including irritable bowel syndrome and multiple inflammatory bowel diseases.
- Chronic stress is associated with reduced diversity in the microbiome and increased intestinal permeability.
- Emerging evidence suggests that our gut microbiota can influence stress responsiveness.
- Implementing stress management techniques can help to address stress-derived digestive issues.
☝️DISCLAIMER☝This article is for informational purposes only. It is not intended to constitute or be a substitute for professional medical advice, diagnosis, or treatment.