Probiotic supplements have grown in popularity, with many consuming both capsules and drinks containing live cultures in the hope of improving their gut health. Find out what probiotics are and whether there is any evidence to support the health claims made about probiotic supplements.
Table of contents:
- What are probiotics?
- Are probiotics safe?
- The hierarchy of evidence in clinical research
- Antibiotics-Associated Diarrhoea
- IBS
- Travellers diarrhoea
- Prebiotics and probiotics
- Summary
What are probiotics?
Probiotics are live bacteria that can confer health benefits to a human host. They are known as “friendly” bacteria because they can compete for space and food in the large intestine, thereby preventing harmful or pathogenic bacteria from colonising the gut.
Probiotics are also able to support the immune system and produce short-chain fatty acids such as butyrate, known to reinforce the intestinal wall and reduce inflammation. They can be found in live fermented foods like kefir, kimchi and sauerkraut as well as in dietary supplements.
In recent years, probiotic supplements have grown in popularity, with many seeking to balance their gut microbiome by consuming live cultures in capsule or liquid form. Despite this, some have questioned the benefits of probiotic supplements, doubting whether sufficient amounts of live cultures make it to the gut and whether they contain enough live bacteria in the first place.
Whilst not all probiotic supplements are made equal, there is a body of evidence suggesting that some strains and species of beneficial bacteria can be helpful in treating certain conditions, most notably, antibiotic-associated diarrhoea (AAD), travellers diarrhoea, ulcerative colitis, clostridium difficile disease and some of the digestive symptoms of IBS, such as bloating, constipation and gas.
The most studied probiotics are Lactobacillus, Bifidobacterium and the yeast, Saccharomyces boulardii.
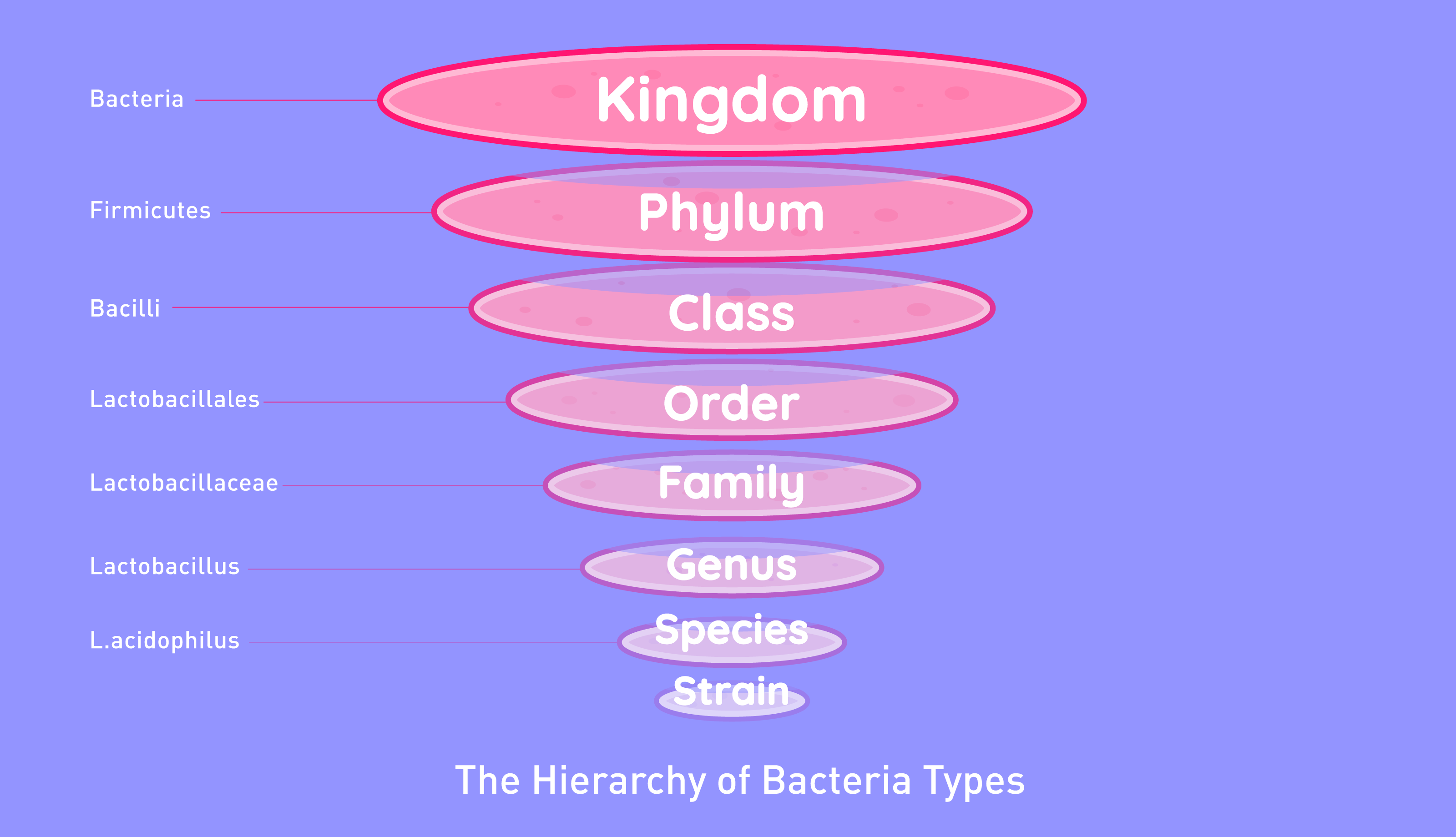
When you see a probiotic on a product label, it will usually be split into three parts: the genus, species and strain. For example, you might see an ingredient such as the following: Lactobacillus acidophilus MN5.
In this case, Lactobacillus is the genus, acidophilus is the species and MN5 is the strain. It should be noted that just because one strain of a genus might have health benefits, it doesn't mean all probiotics of the same genus will do the same.
Are probiotics safe?
Probiotics are considered safe for most healthy individuals, although we recommmend that you consult your doctor before taking any supplementation. In one systematic review looking at harm reporting in Randomised Controlled Trials, researchers concluded that many studies contained insufficent or inadequate discussion on side-effects. Others have raised concerns that probiotics could potentially transfer antibiotic resistant genes to pathogens.,though further research is required.
Lastly, some individuals are at a higher risk for adverse side-effects, including those who are immunocompromised, have a short bowel or suffer with a serious underlying illness. Young infants are also at a higher risk for developing side-effects from probiotics. Anyone in these categories should not take supplementation unless a health professional says otherwise.
☝Did You Know?☝ Probiotics are the most common dietary supplement in the U.S. behind minerals and vitamins
The hierarchy of evidence in clinical research
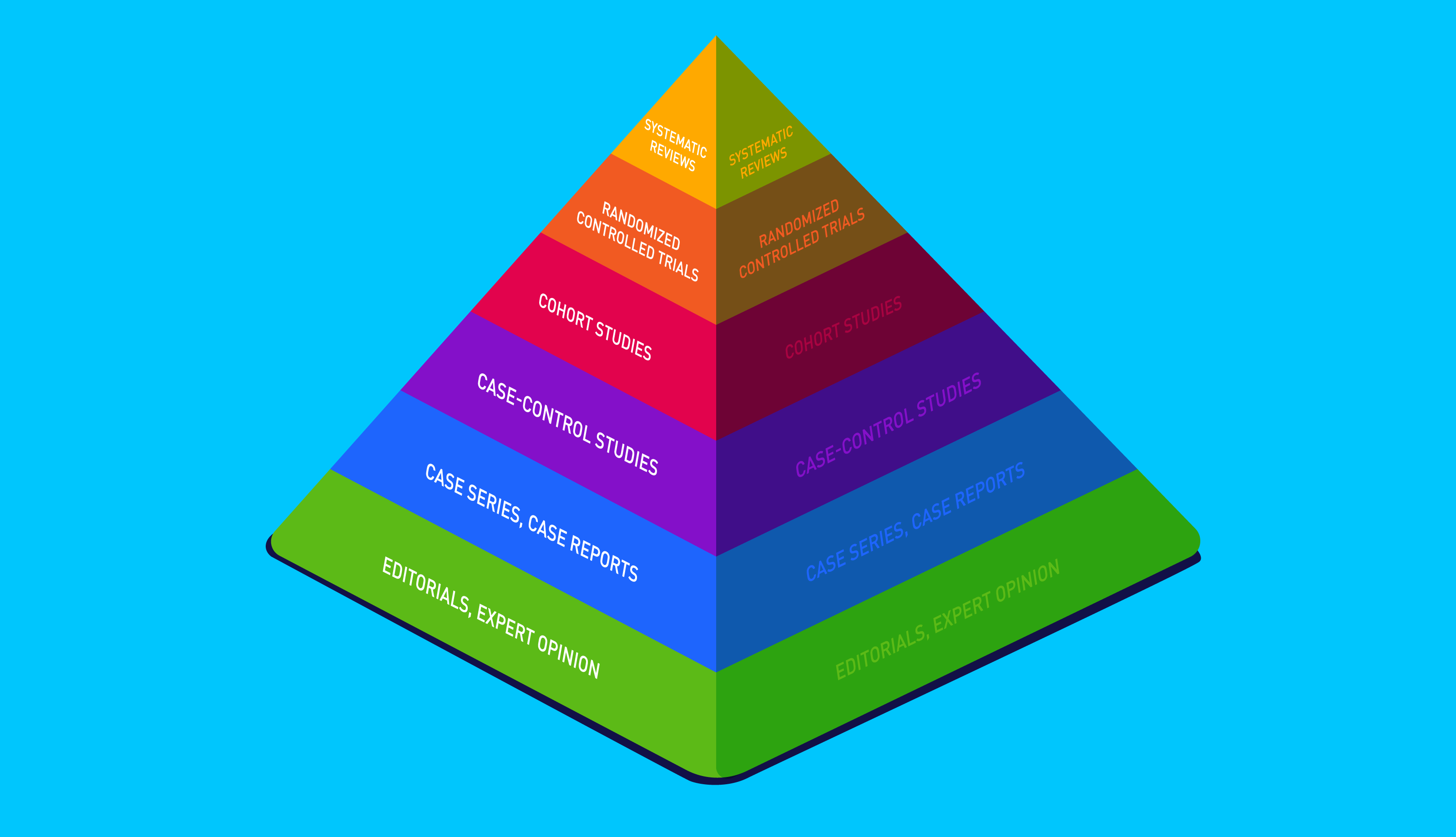
Before we look at the current research on the benefits of probiotics, it is important to take a quick detour to examine the hierarchy of evidence. In short, researchers broadly rank the value of different research methods based on how rigorous they are. At the top of this hierarchy are systematic reviews and meta-analyses, ideally looking at randomised, controlled trials.
It should be noted that not all studies are made equal, with study size and blinding affecting the outcomes. A double-blind study means that neither the researchers nor the participants know who is in the placebo group and who is in the treatment group, whilst a single-blind study means only the participants do not know which treatment they are recieving.
Antibiotics-Associated Diarrhoea
A healthy microbiome should be rich and diverse, containing a stable balance of bacteria to protect against pathogenic species. Multiple factors can affect the composition and richness of our gut bacteria, though poor diet and antibiotics have a particularly pronounced effect.
The microbiome is a living and dynamic ecosystem that is shaped by lifestyle, diet and drugs such as antibiotics.
When we take antibiotics, they do not distinguish between beneficial bacteria and opportunistic bacteria. Think of them like a hand grenade going off in a crowded microbial city. This loss of beneficial bacteria can result in diarrhoea and the vacuum left can allow for gut bugs like clostridium difficile to set up shop.

Though more research is needed, there is a promising body of evidence that some species of probiotics can help to minimise antibiotics-associated diarrhoea (AAD). It is thought that probiotics can recolonise the gut and restore balance after a course of antibiotics, securing the vacant spaces and preventing opportunistic bacteria from populating these.
For example, in a Randomised Controlled Trial (RCT), it was shown that a probiotic formulation containing Lactobacillus acidophilus LA-5 and Bifidobacterium BB-12, when compared to placebo, significantly reduced the duration of AAD, though it did not lower the prevalence of this.
Moreover, in a systematic review and meta-analysis of 11,811 individuals, researchers found a statistically significant association between the use of probiotics and the reduction of AAD. Whilst the majority of the screened studies used lactobacillus, either alone or in combination, the exact strains were poorly documented, preventing more specific conclusions from being made.
In another systematic review and meta-analysis which looked at 17 RCT’s, it was found that AAD was present in around 8% of the probiotic groups compared to 17.7% in the control groups- an almost 50% difference. Likewise, a meta-analysis of 23 studies and over 3000 individuals yielded similar results: the incidence of AAD in probiotic groups was found to be 8% compared to 19% in control groups.
The strains and mixtures of probiotics varied across the studies analysed, with some using single-strain probiotics and others up to 10 strains. This study concludes by suggesting that Lactobacillus rhamnosus or Saccharomyces boulardii at 5-40 billion colony-forming units/day may be an appropriate dose given its effectiveness and the likelihood that bad reactions are rare. In a separate meta-study, Lactobacillus rhamnosus GG was shown to significantly reduce the development of AAD also.
In summary, further research is needed to determine which strains and doses work best. With that being said, a few strains of Lactobacillus and Saccharomyces boulardii show early promise in their ability to prevent ADD or reduce its duration.
Clostridium Difficile Related Diarrhoea (CDAD)
As we have discussed, antibiotics can significantly alter the microbial communities in our colon, challenging the balance of this inner ecosystem. When this happens, gut superbugs like Clostridium Difficile (CD) can move in, leading to severe diarrhoea and other complications.
Though this can be cured with further antibiotics, infection re-occurs in as many as four in ten individuals, likely because the microbiome balance is disturbed again. CD is more common in those who take antibiotics for long periods of time, and by extension, the elderly.
Whilst there is a need for larger-scale studies, research to date has shown that a select few probiotics might be an effective treatment for CD related diarrhoea, though not CD infection more generally.
For example, In one meta-study looking at over 3000 subjects in 25 RCT’s, Saccharomyces boulardii was shown to be effective in reducing the development of CDAD (clostridum difficile related diarrhoea). Likewise, in a separate systematic review looking at 31 RCT’s and including 8672 patients, researchers concluded that probiotics might be an effective way to prevent CDAD. More specifically, the study suggests that when probiotics are taken with antibiotics, the risk of developing CDAD is reduced by as much as 60%.
In an ongoing prospective study, researchers have reported that patients seem to have less recurrent CDAD and quicker recovery whilst taking Lactobacillus GG. With that being said, the researchers cautioned that it is too early to measure statistical significance.
In short, there is some research suggesting that some probiotics can prevent diarrhoea related to the clotridum difficle infection, though it is still unclear which strains, doses and length of treatment are most effective. Furthermore, it must be noted that neither the Food and Drug Adminstration (FDA) nor the Centre For Disease Control (CDC) recommend probioitcs in the treatment or prevention of C.Diff infection. The CDC in particular has stated more research is needed to elucidate the health benefits of Saccharomyces boulardii.
Irritable Bowel Syndrome (IBS)
IBS is a common condition that affects the digestive system. Symptoms include bloating, abdominal pain, constipation and diarrhoea and may come and go over time. It is not known what causes IBS, although the disorder has been linked with dysbiosis in the microbiome. To date, studies suggest that some probiotic strains might be able to alleviate the symptoms of this debilitating disorder. Let's explore a few of these:
Firstly, in a RCT involving 122 individuals, the probiotic Bifidobacterium bifidum MIMBb75 was shown to significantly improve both quality of life and IBS symptoms, including pain, bloating, and urgency. What’s more, the probiotic was well-tolerated and adverse events were no different from the placebo group.
Continuing, a systematic review which screened 11 RCT’s also found evidence for their efficacy in the treatment of some IBS symptoms. Out of 11 studies, 7 revealed that supplementation with probiotics significantly improved IBS symptoms compared with placebo groups. In the remaining four studies, no significant improvement was noted after probiotic supplementation.
Interestingly, all but one of the studies using multi-strain probiotics showed improvements, whereas all the single-strain probiotic studies showed no difference to the placebo group. This suggests that multi-strain probiotics are more effective in treating IBS than single-strain ones. Before we draw any conclusions, another systematic review and meta-analysis published last year suggested the exact opposite, instead noting that single-strain probiotics at a higher dose seem to be the better choice for IBS sufferers.
In clinical research, the most highly regarded study design is a randomized, double-blind, placebo-controlled trial. A study that meets all these critera investigated the effect of a probiotic strain, L. plantarum CCFM8610, on the symptoms of IBS-D (IBS with diarrhoea) in 75 patients.
During the 12-week trial, the control group received oligosaccharides, whilst the study group received 10 billion colony forming units of L. plantarum daily. Although small, the quality of the study makes it a valuable piece of research.
The group taking the lactobacillus strain reported a significant increase in quality of life and a decrease in the severity of their symptoms. Their microbiomes also showed increased diversity, a decreased abundance of the bloating-related genus Methanobrevibacter, and an increase in bacteria that create butyrate, an essential short-chain fatty acid.
To summarise, whilst further research is needed, there is compelling evidence that probiotics can alleviate IBS symptoms and help individuals manage the disorder. Despite this, it is still not clear whether single-strain or multi-strain probiotics are more effective, at which dose/duration they yield the best results and which strain is the best option for IBS sufferers, though L. plantarum has shown early promise.
Travellers Diarrhoea (TD)
Travellers Diarrhoea (TD) is a common digestive issue experienced by those visiting foreign countries, particuarly underdeveloped nations. It is often caused by bacterial infections from contaminated food and water. In comparison to the other conditions mentioned, the number of trials looking at probiotics in relation to TD is low.
In 2007, a meta-analysis looking at 12 RCT’s found that several probiotics (Saccharomyces boulardii and a mixture of Lactobacillus acidophilus/Bifidobacterium bifidum) significantly prevented the development of TD. No serious side effects were reported during the trials.
Despite this, in 2017 The International Society of Travel Medicine published guidelines on treating travellers diarrhoea. After reviewing the evidence for probiotics, they concluded that there is insufficient research to recommend commercial probiotics in the prevention or treatment of TD. The reasons given for this judgement included variations in the doses, strains and geographic destinations between studies.
In response, a 2018 study conducted a meta-analysis on 11 RCT’s, concluding that probiotics had a statistically significant effect on the prevention of TD, though the paper did not assess the quality of the studies it used or measure the prevalence of side-effects.
Overall, despite some evidence suggesting probiotics have a small benefit in those with TD, the research is in its infancy meaning no definitive conclusions can be reached. More large-scale studies are needed before we can recommend any probiotic strain as a treatment for TD.
| Condition | Probiotic with potential benefits in clinical trials |
|---|---|
| Antibiotic Associated Diarrhoea | Lactobacillus acidophilus LA-5, Lactobacillus rhamnosus GG and Bifidobacterium BB-12. |
| Irritable Bowel Syndrome | Bifidobacterium bifidum MIMBb75 and L. plantarum CCFM8610. |
| Travellers Diarrhoea | Saccharomyces boulardii, Lactobacillus acidophilus and Bifidobacterium bifidum. |
| Clostridium Difficile | Saccharomyces boulardii and Lactobacillus GG. |
A list of the probiotics shown to have potential benefits and what conditions the clinical trials looked at
Prebiotics and probiotics
Probiotic supplements are not a panacea, nor will drinking a cultured beverage each morning “fix” your gut health. In fact, you can live a healthy life and optimise your gut health without ever taking a supplement, simply by eating a diet rich in probiotics and prebiotics. Prebiotics foods include artichokes, legumes, beans and plant based fibre, whilst probiotic foods include fermented goods such as:
- kimchi
- kombucha
- kefir
- miso
- sauerkraut
Whilst those with certain conditions might benefit from supplements, Atlas advocate a food first policy and highly recommend increasing plant-based dietary fibre before anything else. This is because a healthy, balanced and diverse diet will give your beneficial bacteria all the fuel they need.
TIP: Take the Atlas Microbiome Test and receive weekly, personalised food recommendations to encourage beneficial bacteria and help cultivate diversity in your gut.
Summary
Although there is some research suggesting that specific probiotic strains may help treat IBS symptoms and infectious diarrhoea, this is not sufficient to make conclusive statements at the moment.
Continuing, there is no clear consensus on which strains, doses and mixtures of probiotics are most effective. In the absence of more specific research, we simply cannot make concrete health claims or recommend supplements as a treatment for certain conditions.
As a result, we recommend that you increase probiotics through dietary changes instead of dietary supplements.
☝️DISCLAIMER☝This article is for informational purposes only. It is not intended to constitute or be a substitute for professional medical advice, diagnosis, or treatment.