The gut microbiome does far more than just digest fibre. Discover how bacteria are involved in inflammatory bowel disease and common metabolic diseases.
Learn how gut microbes can influence your cardiovascular and digestive health, and protect you from obesity, atherosclerosis, diabetes type 2, Crohn’s and ulcerative colitis.
It just so happens that there are specific specific trends and patterns in the emergence of these diseases, even though some don’t seem remotely linked to gut bacteria at all.
As you can see, we're not just talking about digestive conditions, but metabolic ones too. This is evidence that the activity of your gut microbes can affect any part of your body, not just the colon.
We use data from scientific research in our microbiome test, and we thought you might like to know how it works. But first, a short list of the illnesses we'll be discussing.
| Metabolic diseases | Atherosclerosis |
| Diabetes type 2 | |
| Obesity | |
| Inflammatory bowel disease (IBD) | Crohn's disease |
| Ulcerative colitis |
Whether you've taken our microbiome test or not, this article will brief you on what you need to know on how bacteria protect you from 5 serious chronic diseases.
These illnesses were hand-selected by our team of microbiome researchers as having the strongest evidence of reliably specific gut microbiome features.
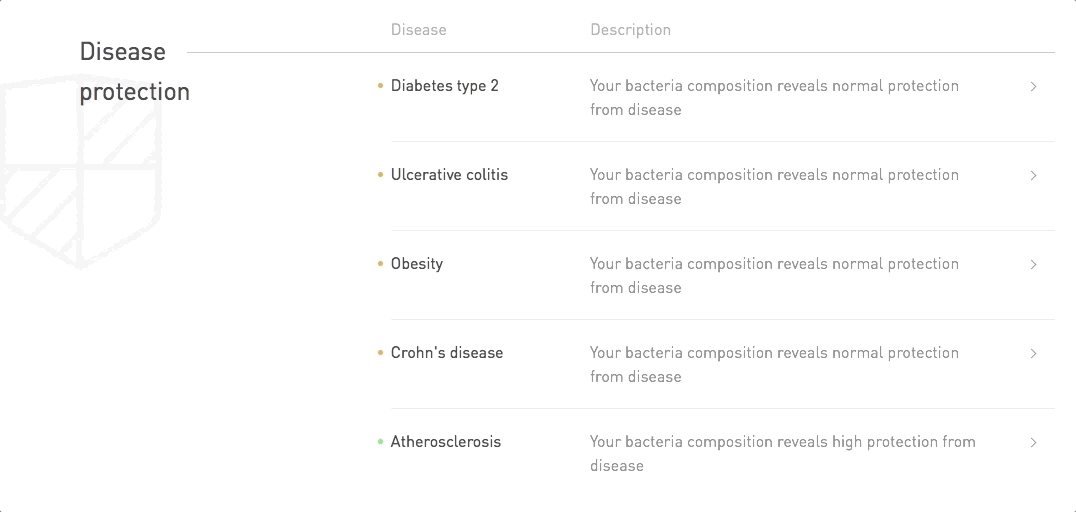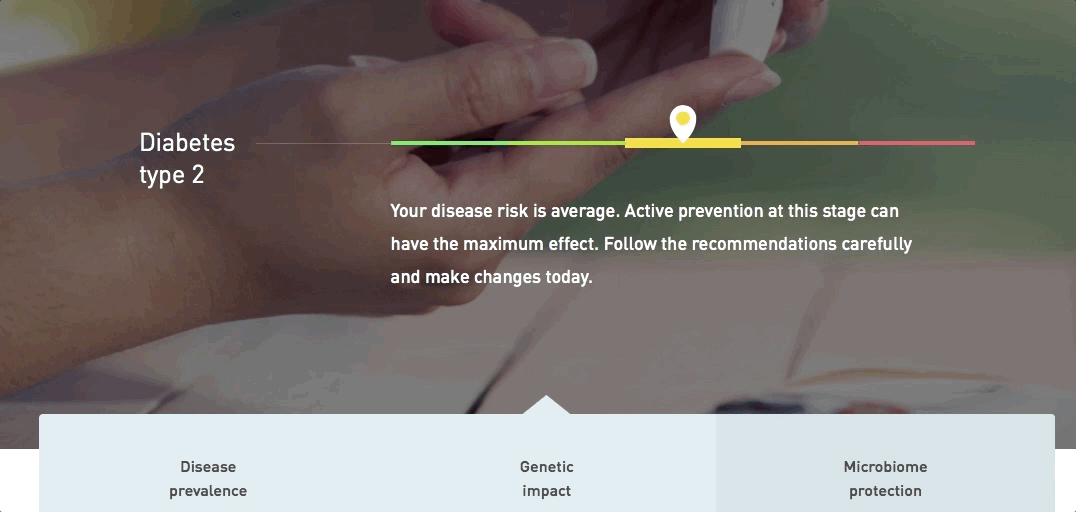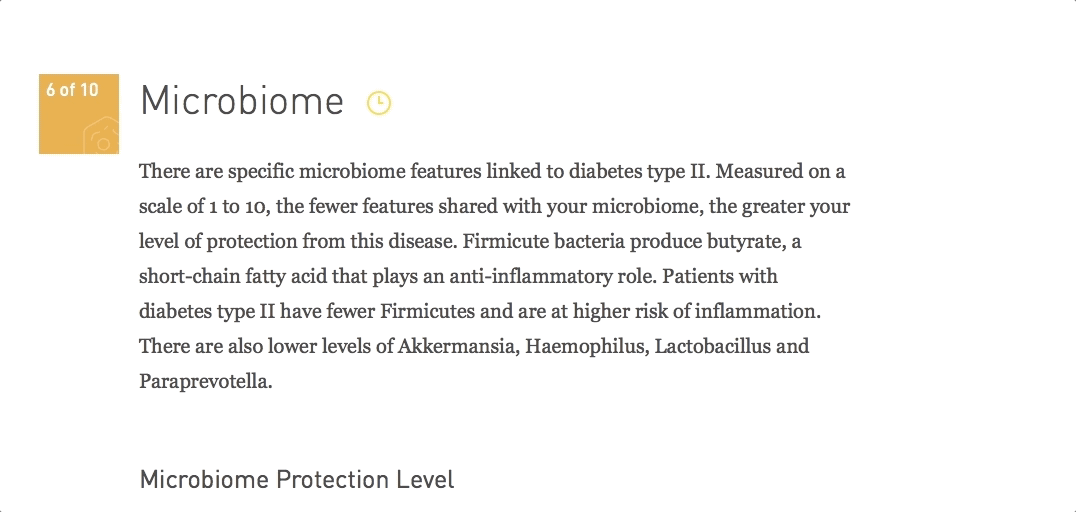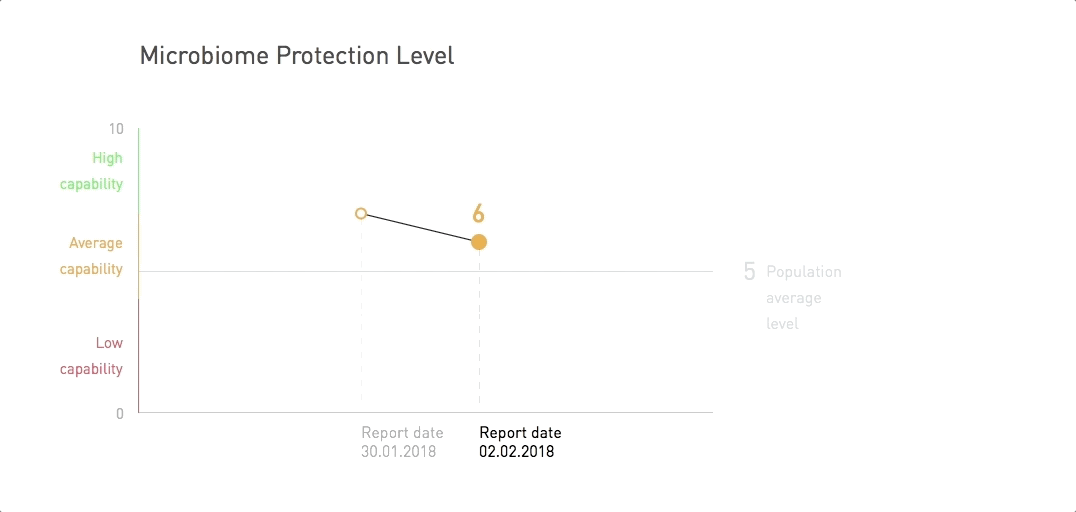
Disease protection conferred by bacteria is assessed based on how similar or different your gut microbes are to the gut microbiome of patients with this disease.
☝️TIP☝️ If you've taken the Atlas Biomed Microbiome Test, you can access your disease protection profiles by clicking on the links in the sections below.
Metabolic diseases
When doctors talk about metabolism, they are referring to the chemical reactions that occur naturally within the body to maintain life.
Important metabolic processes include the breakdown (catabolism) of fats, proteins, carbohydrates and nutrients as well as their transformation (synthesis) into compounds that keep the body well-oiled, fueled and healthy.
When abnormal reactions happen in the body, they can lead to problems processing essential compounds that can accumulate excessively in bodily tissues and organs or, on the contrary, create deficiencies.
As evidence grows in this field, medical research suggests that atherosclerosis, obesity and diabetes type 2 are all linked by more than just the metabolism, by the gut microbiome too.
Atherosclerosis
Colloquially referred to as "hardening of the arteries" and nicknamed the "silent killer", atherosclerosis is a serious condition in which plaque builds up in the arteries.
Atherosclerosis is associated with lower levels of Akkermansia muciniphila, a microbe found in lean people, Roseburia and Eubacterium that produce an important nutrient called butyrate, and Dorea.
The gut microbiome of patients also show increased levels of other bacteria like Collinsella and Prevotella.
In particular, patients with athersclerosis have higher levels of Proteus, Edwardsiella and Providencia that produce trimethylamine (TMA) from choline (found in egg yolks) and L-carnitine (found in red meat and fish).
Several studies indicate that this disease, and several others, have markers of a more inflammatory gut environment than healthy controls, the participants used to compare the findings of researchers.
| ATHEROSCLEROSIS | Go to my disease protection report now |
Diabetes type 2
Diabetes mellitus type 2 (T2D) is a metabolic condition in which the body becomes insulin resistant: the system responsible for collecting up excess glucose in the bloodstream to maintain healthy blood sugar levels doesn't work properly.
Excess circulating glucose (hyperglycaemia) damages the blood vessels, tissues, nerves and organs. In particular, people with diabetes type 2 are at significant risk of complications including heart disease, stroke, kidney disease, nerve and eye damage.
It is widely suggested that the increasing popularity of the Western diet, high in fats and sugars, is partly responsible for the increase of inflammatory diseases, like T2D. This is because it is low in dietary fibres that nourish the microbiome, affecting the health-promoting functions they perform.
Studies comparing the gut microbiome of T2D patients and healthy controls show dysbiosis, negative alterations in the composition of the gut microbiome. Dysbiosis favours abundance and presence of bacterial types that affect the human host’s health.
In particular, members of the Firmicutes phylum known for synthesising this nutrient were less abundant, like Faecalibacterium prausnitzii, Roseburia and Eubacterium.
There are also lower levels of Lactobacillus, a probiotic bacteria that also has anti-inflammatory properties and Akkermansia, a common microbe detected in lean people that is known to protect against insulin resistance and metabolic conditions.
| DIABETES TYPE 2 | Go to my disease protection report now |
Obesity
Genetics is a risk factor for obesity, but environment, social and lifestyle factors are important predictors in determining whether a person is more likely to develop this condition.
These include following a “Western diet” of high-fat, high-sugar, high-processed foods; sedentary living and lack of exercise as well as coming from a deprived social background.
Research into the gut microbiome of obese patients shows that, overall, there is less diversity of microbes in the gut. Studies show increased abundance of Bacteroides and Parabacteroides that thrive on Western diet-type foods.
Results also show the presence of Enterobacteriaceae and Desulfovibrionaceae that are associated with metabolic problems.
When there are too many or too few of the same bacteria, or elevated presence of rare opportunistic bacteria, it indicates dysbiosis, negative alterations in the microbial ecosystem that contributes to inflammation and illness.
Obesity is also linked to lower levels of Akkermansia muciniphila, like in diabetes type 2. A. muciniphila is associated with lower body mass, protection from insulin resistance and metabolic disorders.
| OBESITY | Go to my disease protection report now |
Inflammatory bowel disease (IBD)
The inflammatory bowel disease group covers Crohn’s disease (CD) and ulcerative colitis (UC). Both of these conditions are characterised by acute episodes of inflammation (flares) that affect the digestive tract.
In addition to nutrient malabsorption and internal scarring, people with IBD are at higher risk of several health problems including colorectal cancer, blood clots and primary sclerosing cholangitis, severe inflammation that affects the bile ducts.
While genetic predisposition increases a person’s risk of getting IBD, there is now a lot of research demonstrating the influence of environmental and lifestyle factors.
Mounting evidence indicates that dysbiosis, negative alterations in the gut microbiome, can cause abnormal immune reactions that lead to chronic inflammation. This is believed to increase the risk of becoming ill for people with genetic risk traits.
☝️IMPORTANT☝️ The microbiome of a patient with IBD in remission can look very similar to that of a healthy person. That is because a microbiome test is just one snapshot of the gut microbiome at a specific point in time. This ecosystem is very susceptible to change, and that is why it can look very different when a patient is experiencing a flare.
Crohn’s disease
Any part of the digestive tract from the mouth to the anus can be affected by inflammation in Crohn’s disease. The lesions occur on patches of the gut lining and can penetrate every layer of it.
Diarrhoea, bloody stools, mouth ulcers, fatigue, anaemia, weight loss and loss of appetite are symptoms of acute inflammation, known as flares. There is no cure for Crohn’s and the healthcare aim is long-term remission.
Dysbiosis, lower diversity and higher levels of normally rare bacteria with pathogenic traits are common in the microbiome of Crohn’s patients.
For example, increased abundance of certains types of Escherichia coli , pro-inflammatory bacteria that adhere to the gut lining has been identified by several studies.
At the same time, there are significant differences between the microbiome of someone with Crohn’s disease who is experiencing a flare and a patient in remission.
Studies of patient microbiomes undergoing a flare in inflammatory activity have reduced levels of Faecalibacterium prausnitzii, bacteria of the Firmicutes phylum known for their butyrate-producing capacities.
| CROHN'S DISEASE | Go to my disease protection report now |
Ulcerative colitis
Inflammation is not patchy, but continuous, usually starting at the rectum and spreading into the colon. Damage only affects the innermost layer of the gut lining, called the mucosa.
Symptoms of a flare include rectal pain and bleeding, urgent need for defection, pain and diarrhoea with blood or puss. There is no cure for ulcerative colitis and the healthcare aim is long-term remission.
According to studies, dysbiosis is common in the gut microbiome of patients with UC. Researchers have noted more abundance of pathogenic bacteria like Escherichia coli, Clostridium difficile, Helicobacter and Salmonella.
These bacteria are all associated with infections, including gastroenteritis, and several studies have also linked specific infections by these bacteria to increased risk of developing UC.
There are also trademark features in patterns of commensal bacteria with health-promoting functions, like the production of butyrate. Patients with this disease tend to have less Faecalibacterium, Eubacterium, Ruminococcus, Akkermansia and Phascolarctobacterium.
| ULCERATIVE COLITIS | Go to my disease protection report now |
So how do you calculate disease protection?
Based on the same techniques and methods as researchers, we use 16s rRNA genetic sequencing to identify the quantity and relative abundance of bacteria in your microbiome.
Then we compare it to the scientific evidence and assess whether your microbiome composition is closer or further from the trademark features and patterns associated with each disease.
Our team of scientific researchers have also dedicated an important part of the results to tailored food recommendations based on your bacteria. These are designed to help you improve the composition of your gut microbiome, enhancing disease protection and nutrient synthesis.
By following weekly personalised eating recommendations, you can increase the abundance of beneficial species that are involved in maintaining a stable and healthy microbiome environment.
In the future, microbiome research will not only identify patterns in the make-up of the gut microbial ecosystem, but possibly find new means to diagnose and treat disease too.
It is believed that at-home testing like the Atlas Biomed Microbiome Test can help people identify health risks in the composition of their microbiome and take small steps to improving their wellbeing before disease strikes.
- S. Banquera et al., Global Overview of the Epidemiology of Atherosclerotic Cardiovascular Disease, 2015
- J. Zhuye et al., The gut microbiome in atherosclerotic cardiovascular disease, 2017
- C. Bogiatzi et al., Metabolic products of the intestinal microbiome and extremes of atherosclerosis, 2018
- Type 2 Diabetes health problems, NHS information page
- ME Cerf, Beta cell dysfunction and insulin resistance, 2013
- J Qin et al., A metagenome-wide association study of gut microbiota in type 2 diabetes, 2012
- W.Aw and S. Fukuda, Understanding the role of the gut ecosystem in diabetes mellitus, 2018
- NHS conditions, obesity summary
- CD Davis, The gut microbiome and its role in obesity, 2017
- RE Ley et al., Obesity alters gut microbial ecology, 2005
- E Le Chatelier, Richness of human gut microbiome correlates with metabolic markers, 2013
- NA. Molodecky et al. Environmental Risk Factors for Inflammatory Bowel Disease
- NA. Molodecky et al., Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review, 2012
- What is Inflammatory Bowel Disease, Center for Disease Control
- Managing flares, Crohn’s and Colitis Foundation of America
- The Lancet Crohn's Disease Review
- J E Mawdsley and D S Rampton, Psychological stress in IBD, 2005
- S. Khanna & LEH Raffals, The Microbiome in Crohn's Disease. Role in Pathogenesis and Role of Microbiome Replacement Therapies, 2017
- V. Pascal et al., A microbial signature for Crohn's disease, 2017
- Ulcerative colitis overview, National Institute for Diabetes, Digestive and Kidney Diseases
- E Valera et al., Colonisation by Faecalibacterium prausnitzii and maintenance of clinical remission in patients with ulcerative colitis, 2013
- NR Bullock et al., Comparative composition of bacteria in the human intestinal microflora during remission and active ulcerative colitis, 2004
- ZH Shen et al, Relationship between intestinal microbiota and ulcerative colitis: Mechanisms and clinical application of probiotics and fecal microbiota transplantation, 2018
- K. Machiels et al., A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis, 2014