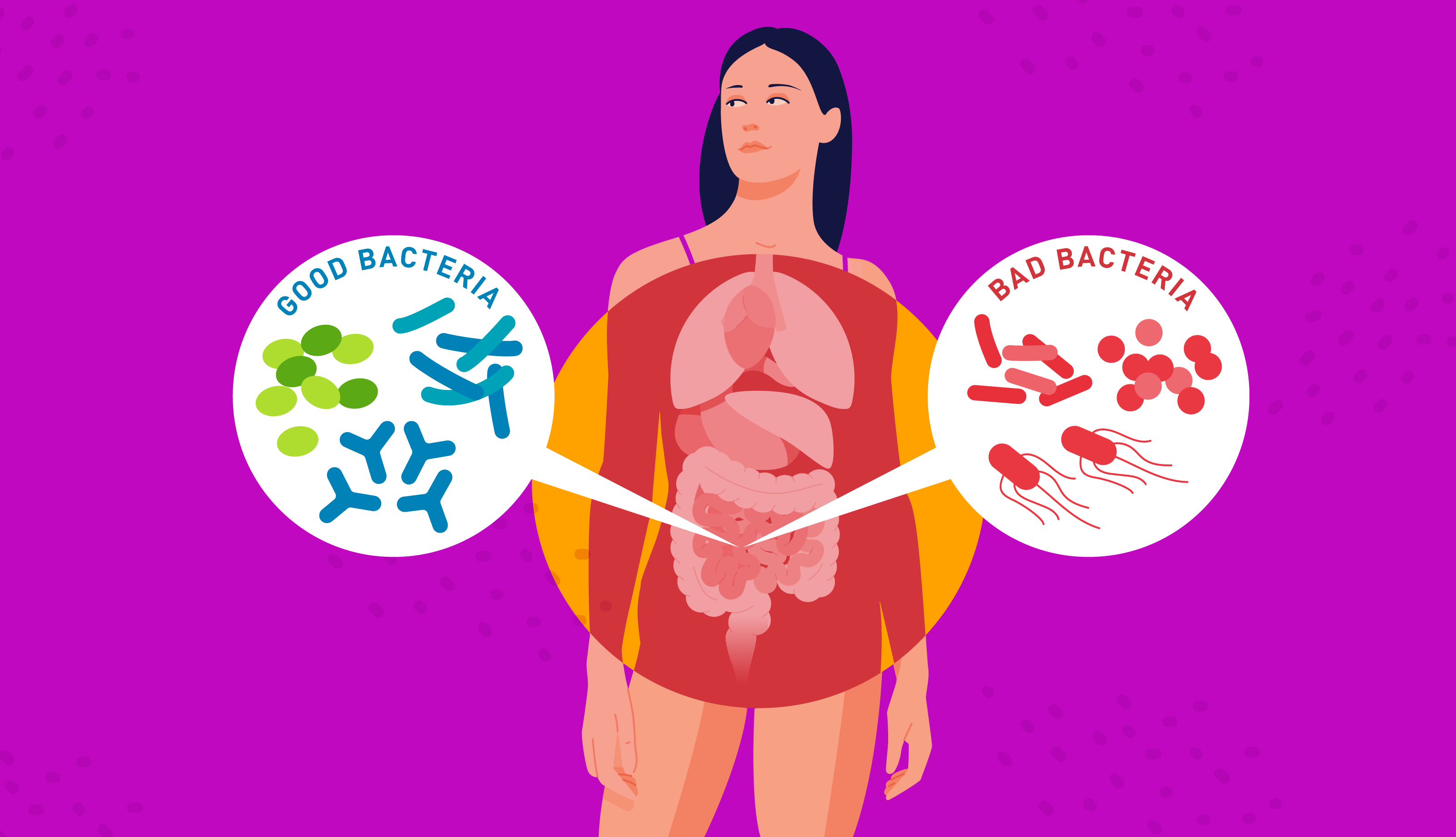
Imbalances in your gut microbiome can cause chaos for your digestion, health, and wellbeing. It’s known as dysbiosis, and your lifestyle could be a major influence.
Your gastrointestinal tract is a major interface between the inside of your body and the outside world. On average, you will consume around 60 tons of food in your lifetime - all of which will pass through it and be digested, nutrients absorbed, and waste products will exit via the back door.
This means your gut will be exposed to food, dietary toxins, opportunistic microbes, and medication, which have the potential to alter the composition and diversity of your gut microbiota: an ecosystem of bacteria that reside in your colon.
Table of contents
- 1. Your birth and beyond
- 2. Antibiotics don’t discriminate
- 3. A diet low in fiber
- 4. The Western diet downfall
- 5. Chronic disease disruption
- 6. Less exercise, less microbes
- 7. Low-carb diet deception
- 8. Stress hurts the gut too
- 9. Prescription drug pitfalls
- 10. The microbiome ages too
- 11. Travel and Delhi belly
Most gut bacteria live in harmony with you: you give them with shelter and food, and in exchange, they provide you with several health benefits. But if you have too much of one microbe, not enough of another, or there is a lack of diversity, your health can be affected. This is dysbiosis.
Dysbiosis doesn’t just occur, external factors affect the composition of your gut microbiome. Although it is unique to you, certain aspects of your lifestyle can disturb its harmony, including the way you were born, your diet, activity levels, and your exposure to antibiotics.
1. Your birth and beyond
Whether you were born by vaginal birth or C-section can influence your gut microbiota from the day you enter this world.
If you were delivered naturally, microbes colonised your gut as you were born. Because as you were welcomed into the world, you were baptised by an array of your mother’s vaginal and fecal bacteria. Sounds gross, right? But it helped to build your gut into what it is today.
Delivery by caesarean section can delay the establishment of gut microbes, like Bifidobacterium and Lactobacillus, because of the sterile techniques used. It also means these babies are more susceptible to developing allergies and asthma. In short, your gut microbiome and immune system are linked.
Exposure to certain bacteria during birth helps develop your immunity and lays the foundations for the strength of your microbiome through life. But don’t sweat the small stuff over how you entered the world, there’s plenty of time for your gut to catch up if needs be. Rome wasn’t built in a day, as they say.
2. Antibiotics don’t discriminate
Antibiotics can shape the composition and diversity of your gut microbiome. And, they have been linked with making long-lasting changes to it.
Your gut microbiome is constantly adapting to changes in you and your environment to keep your intestinal environment stable. But treating infections with antibiotics disrupts your microbial community. Ultimately, this can reduce the abundance of probiotic microbes like Bifidobacterium and Lactobacillus.
The gut microbiome is a rainbow of functions and bacteria
After all, antibiotics are used to treat bacterial infections, and they don’t discriminate. By eliminating many microbes, it can leave room for opportunistic microbes to grow out of control, causing digestive distress. The over-prescription of antibiotics in recent decades has only added to the problem.
Overexposure to antibiotics can have long-term consequences for the diversity of your intestinal microbiome. It can take several months and even years for this ecosystem to recover after antibiotic use, which is why they should be used with caution and only when your doctor recommends them.
3. A diet low in fiber
Your gut microbes love complex carbs, and by breaking them down, they encourage the growth of other friendly bacteria in your gut. Without fiber, your gut microbiome lacks variety.
It’s true, eating plenty of dietary fiber influences the composition of your gut microbiota and is linked to better health. Gut microbes like Bifidobacteria and Lactobacilli break down complex carbohydrates through fermentation, and turn them into short-chain fatty acids (SCFAs) like acetate.
Prebiotic fibers and acetate produced by other bacteria are used by beneficial microbes that produce a short-chain fatty acid called butyrate. Butyrate is particularly useful because it is the main source of energy for the special cells lining your gut, called colonocytes. It even prevents inflammation and gut dysbiosis.
The be all and end all here is if you want a healthy microbiome, your gut needs fiber. Dietary fiber is found in many plant-based foods, and their ability to sustain the trillions of bacterial cells in your gut makes them prebiotics.
Foods which help to increase the abundance of specific bacteria
| Bacteria | Food Sources | SCFAs produced |
|---|---|---|
| Bifidobacteria | beetroot, rye, barley, oats, onions, garlic, Jerusalem artichokes | acetate, lactate |
| Lactobacillus | rye, wheat, onions, garlic. chicory, milk, legumes | lactate, acetate, butyrate, propionate |
| Butyrate-producing bacteria | barley, oats, wholegrains, rye, apples, onions, mushrooms, garlic | butyrate |
4. The Western diet downfall
The classic western diet has been criticised for its lack of nutritional value, and it has your gut microbes in turmoil too.
The Western diet is characterised by a high intake of meat, fat, and refined sugar. And if that wasn’t enough, the diet is seriously low in fiber content too. Overall, the Western diet affects many aspects of your health, including chronic disease risk, your gut, and its bacteria.
There’s plenty of research confirming changes to the gut microbiota associated with obesity and metabolic diseases that are linked to the Western diet. It also affects the integrity of your gut lining where a less diverse environment may result in inflammation and a disruption to the gut homeostasis.
When it comes down to it, the Western diet is SAD. Pardon the pun, but that’s also what medical researchers call it: S-A-D, the Standard American Diet. The high-fat content increases gut permeability and the risk of inflammation - situations which may lead to the growth of opportunistic bacteria, reduction in healthy bacteria, inflammatory responses, and/or chronic disease.
5. Chronic disease disruption
A balanced microbiome can keep you healthy, and patterns of dysbiosis have been detected in many common chronic metabolic and inflammatory diseases.
Health-promoting bacteria like Bifidobacteria, Lactobacillus, Akkermansia, and Faecalibacterium are often less abundant in individuals with a chronic disease and other bacteria, including opportunistic microbes, may be present at elevated levels.
Your gut microbes can have an impact on weight gain, as well as important health markers like blood glucose, cholesterol, and triglyceride levels. These markers are all associated with deadly metabolic diseases that are increasingly common, like diabetes, heart disease, and obesity.
Dysbiosis may also leave the gut more susceptible to infections and make the immune system more reactive, which is shown to play a role in autoimmune illnesses like inflammatory bowel disease, better known as Crohn’s disease and ulcerative colitis.
6. Less exercise, less microbes
If you want a diverse, enriched, and happy gut microbiome, you need to get your sweat on, because like you, your bacteria thrive on exercise .
It’s true. Sedentary people tend to have a less diverse microbiome than people who are physically active. Importantly, physical activity increases the abundance of beneficial bacteria in your gut, enriches the diversity of the microbiome, and can enhance the production of beneficial compounds like SCFAs and anti-inflammatory metabolites.
Nature and exercise are good for gut bacteria and mental health
Exercise, even at a low dose but continuously adhered to, increases the abundance of health-promoting bacteria such as Bifidobacterium, A. muciniphila, and F. Prausnitzii. Cardio workouts, aka aerobic exercise, are best. This includes activities like jogging, walking, cycling, dancing, and swimming. So, what are you waiting for?
Enriching the diversity of your microbiome through exercise (or otherwise) is a great way to keep dysbiosis at bay. Increasing healthy bacteria means your gut barrier functions are improved, inflammation remains low, and risk of illness is reduced.
7. Low-carb diet deception
Low-carb diets are all the rage, but cutting out essential dietary fibers and plant nutrients limits the food sources available to your beneficial gut bacteria.
A low-carb or ketogenic diet insists that the follower has a carbohydrate intake of less than 50g per day. They’ve grown in popularity thanks to the media and celebrity advocates. While in the short term they can have some health benefits, cutting out a food group can reduce the diversity of your gut microbiome.
Plant-based carbohydrates like those present in fruits, vegetables, nuts, and wholegrains provide essential nourishment for beneficial gut microbes. The sustenance provided by these complex carbs means the bacteria can perform their important functions, keep your gut microbial environment diverse, and provide you with numerous health benefits.
Although short-term effects of such diets may be good, in the long-run you could end up with an imbalanced gut. Other research also suggests that long-term low-carb diets increase the risk of dying from heart problems, stroke, and cancers.
8. Stress hurts the gut too
Psychological stress is implicated in the alteration of your gut microbiome. If you’re feeling stressed, some of your beneficial microbes may be affected.
In short, stress is linked to changes in both abundance and types of gut bacteria. And, with the gut and brain having their own communication system, your gut can have profound effects on your mood.
There is lots of research which suggest that when we are exposed to stress, the abundance of the health-promoting bacteria, Lactobacillus, declines. Bifidobacteria abundance is also impacted, while increases in pathogenic microbes, like E. coli, can occur.
The reduction in diversity and the low abundance of beneficial microbes in the gut can influence our mood, cognition, and behaviour. The two-way communication system, known as the gut-brain axis, really is a powerful thing.
9. Prescription drug pitfalls
Drugs like non-steroidal anti-inflammatories, statins, antipsychotics, and painkillers can all alter the architecture of your gut microbiome.
The use of prescription drugs is on the rise. Although they may be given for one illness, the changes they induce in the microbiome can result in gut dysbiosis. In particular, the relative abundance of certain gut bacteria can change.
That means some prescription drugs can give you unpleasant side effects. Opioids, for example, like morphine, are associated with severe constipation. One study found up to 25% of 835 non-antibiotic drugs suppressed the growth of at least one bacterium.
Although more research needs to be done to establish the link between the gut microbiome, drugs, and overall health, certain medications have the potential to affect the gut microbiome, and vice versa.
10. Ageing and the microbiome
Just like your birth, particular lifestages can influence your gut microbiome. And, getting older is no exception.
By the age of 60, like many things, your microbiome starts to look very different to how it did when you were younger. The abundance of protective microbes like Bifidobacteria and Lactobacillus declines, but that of opportunistic microbes, like enterobacteria, increases .
Diet is a major influence as we age. Beige foods like white bread and potatoes are more palatable, and the tea and toast diet is often synonymous with older generations. But these lack dietary fiber essential for our gut microbes to survive and function.
Add to this, reduced mobility, limited exercise, chronic diseases, and prescription medication to manage other health problems, and this may help explain why the elderly gut microbiome is less diverse and more susceptible to infection.
11. Travel and Delhi belly
Holiday time is the perfect excuse to let your hair down. Junk food, alcohol, late nights, and dodgy water can make you the perfect Delhi belly candidate.
Airplanes, changing time zones, eating airport food and local cuisine, lack of hydration, and even stress can disrupt your microbiome’s balance and increase the risk of opportunistic bacteria setting up camp.
Delhi belly, or traveller’s diarrhoea, can make you feel pretty rough. Loose, liquid stools, cramping, and abdominal pain is not how you should spend a holiday. Eating the right foods, particularly those that are plant-based, can increase the abundance of healthy bacteria in your gut.
Eating at the usual time of day is also important because your gut microbes will be expecting it - making them wait can make them confused. You also need to remember to stay hydrated, with water rather than beer. Eating the right foods, drinking water, and staying active goes a long way in ensuring you have a stress and illness free holiday.
Remember this
Dysbiosis is not cool. It occurs when the ecosystem of microbes living in your colon is disrupted. The bacteria in your gut perform essential tasks for your body, like breaking down fiber, producing vitamins, and supporting your immune system.
But dysbiosis can affect these functions. And both its development and progression are impacted by your lifestyle. Some things you have no control over, like the way you were born, but others, like diet and exercise, you do.
Incorporating more fiber into your diet to nourish your health-promoting bacteria, and taking part in some cardio-based activities can positively influence your gut microbiome. Even taking steps to de-stress or remove yourself from stressful situations is a great step to a healthy gut.
☝️TIP☝️Discover your gut bacteria status with the Atlas Microbiome Test and get 10% off when you sign up for blog updates!
- Bressa, C et al. , 2017. Differences in Gut Microbiota Profile Between Women with Active Lifestyle and Sedentary Women. PLOS One: 12.
- Conlon, M, A and Bird, A, R., 2015. The Impact of Diet and Lifestyle on Gut Microbiota and Human Health. Nutrients: 7, pp 17-44.
- Cox, A, J et al., 2014. Obesity, Inflammation, and the Gut Microbiota. Lancet Diabetes Endocrinol.
- DeGruttola, A, K et al. (2016). Current Understanding of Dysbiosis in Disease in Human and Animal Models. Inflamm Bowel Dis: 22, pp1137-1150.
- Dominguez-Bello, M, G et al. , 2010. Delivery Mode Shapes the Acquisition and Structure of the Initial Microbiota Across Multiple Body Habitats in Newborns. PNAS: 107, pp 11971-11975.
- Foster, J, A et al. , 2017. Stress and the Gut-Brain Axis: Regulation by the Microbiome. Neurobiology of Stress: 7, pp 124-136.
- Hawrelak, J, A and Myers, S, P. , 2004. The Causes of Intestinal Dysbiosis: A Review. Alternative Medicine Review: 9.
- Karl, J, P et al. , 2018. Effects of Psychological, Environmental and Physical Stressor on the Gut Microbiota. Front Microbiol: 9.
- Le Bastard et al., 2017. Systematic Review: Human Gut Dysbiosis Induced by Non-Antibiotic Prescription Medications. Alimentary Pharmacology and Therapeutics.
- LeBlanc, J, G et al. , 2017. Beneficial Effects on Host Energy Metabolism of Short-Chain Fatty Acids and Vitamins Produced by Commensal and Probiotic Bacteria. Microb Cell Fact: 16.
- Monda, V et al. , 2017. Exercise Modifies the Gut Microbiota with Positive Health Effects. Oxid Med Cell Longev.
- Nature. , 2018. Gut Microbes are Vulnerable to Wide Range of Drugs. Microbiology.
- Palleja, A et al. , 2018. Recovery of Gut Microbiota of Healthy Adults Following Antibiotic Exposure. Nature Microbiology: 3, pp 1255-1265.
- Valdes, A, M et al. , 2018. Role of the Gut Microbiota in Nutrition and Health. BMJ: 361.
- Zinӧcker, M, K et al. , 2018. The Western Diet-Microbiome-Host Interaction and Its Role in Metabolic Disease. Nutrients: 10.